Cell force microscopy
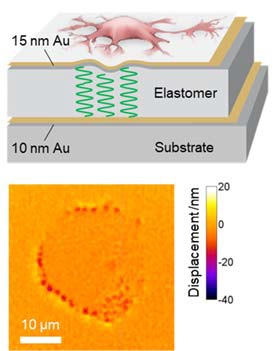
Mechanical forces are at the heart of many processes in biology, e.g. in cell growth, tissue formation, wound healing, the invasion of cancer cells into healthy tissue, and many other diseases. Currently used methods to measure cell forces include micro-patterned substrates and traction force microscopy, but these are often disruptive, require a reference and struggle to provide accurate information on vertical forces (particularly if the forces that cells apply are small).
Expanding on the original cell laser concept, we developed an interferometric method to study the mechanical forces exerted by cells with unprecedented sensitivity [Nature Cell Biology, 2017]. The method was dubbed “Elastic Resonator Interference Stress Microscopy (ERISM)” and can continuously map the mechanical forces that living cells apply while they migrate, divide, and probe their environment. In contrast to all previously described methods, the new technique records high-resolution images of piconewton forces without perturbing the cells or requiring reference measurements and can record force maps over very long periods of time - i.e. many days.
ERISM is particularly attractive in mechanobiology for the emerging area of vertical forces (depth-resolved force imaging), and it is used by us to look at a number of open biological questions related to these. For instance, we have investigated the force dynamics of invadopodia, which are micrometre sized protrusions involved in the invasion of cancer cells and for which it was previously impossible to resolve the mechanical forces they apply [Science Advances, 2020]. We also investigated mechanical aspects of axonal differentiation [Biophysical Journal, 2018], looked at changes in the forces exerted by podocytes during kidney injury [Science Advances, 2018], and analysed the role of different proteins on the cell mechanical phenotype of different human cells [Frontiers in Cellular Neuroscience, 2020; publication in preparation].
Considering mechanobiology more broadly, we combine ERISM with other methods in order to develop a multi-modal mechanical phenotyping system, e.g. with atomic force microscopy (AFM) to investigate cell stiffening as a response to mechanical queues from the environment [Nature Materials, 2020].
Very recently, we have developed a new variant of ERISM, the wavelength alternating resonance pressure microscopy (WARP), which can acquire force maps at dramatically increased acquisition speed and thus allows real time video rate cell force imaging, e.g. of beating cardiomyocytes [Nature Communications, 2021].
